Topics: News, World News, Health
A new drug has been found to slow the progression of Alzheimer's, with some experts calling it a 'turning point' in the fight against the disease.
A study published in the Journal of the American Medical Association found the use of donanemab slowed 'clinical decline' by up to 35 per cent.
The study also found the drug allowed those diagnosed to continue performing daily tasks with independence, such as managing their finances, taking medication, shopping or cleaning.
Alzheimer’s Research UK said the results were symbolic of 'a new era where Alzheimer’s disease could become treatable'.
Advert
The trial, known as TRAILBLAZER ALZ-2, involved almost 1,800 people with early-stage Alzheimer’s and saw half the participants receive a monthly infusion of donanemab and half receive a placebo over the course of 18 months.
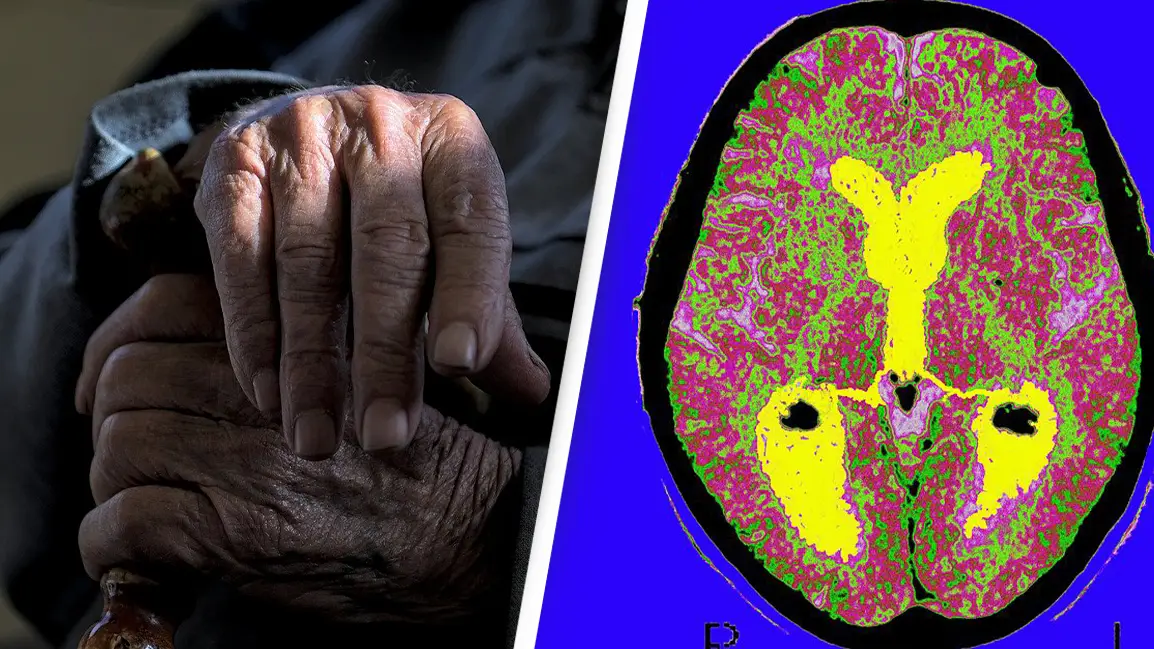
According to the report, the brain scans among the group who received the drug demonstrated low or medium levels of tau, a protein that stabilises the internal skeleton of nerve cells in the brain.
The results of the study were presented at the Alzheimer’s Association International Conference in Amsterdam.
“This is truly a turning point in the fight against Alzheimer’s and science is proving that it is possible to slow down the disease," said Dr Richard Oakley, associate director of research and innovation at Alzheimer’s Society.
“Treatments like donanemab are the first steps towards a future where Alzheimer’s disease could be considered a long-term condition alongside diabetes or asthma – people may have to live with it, but they could have treatments that allow them to effectively manage their symptoms and continue to live fulfilled lives.
“Today’s full results support what we heard about donanemab back in May, that the drug is able to slow down the progression of Alzheimer’s disease by more than 20 per cent.
“This study adds to the growing evidence that treating people as early as possible may be more beneficial, with the effects of donanemab greater in people who were at an earlier stage of the disease.”
A small number of people who took part in the study did experience serious side effects including brain swelling. Furthermore, three deaths in the donanemab group, and one in the placebo group, were considered 'treatment related'.
Dr Oakley also acknowledged that further trials needed more diverse participants.
“We should also note that the majority of people who took part in this trial were white – it’s crucial that in future trials we see more diversity to prove that new drug treatments have similar effects for everyone living with Alzheimer’s disease,” he added.
“Just as we’ve seen a transformation in cancer treatment in recent decades, we’re really hopeful we’re on the same path for dementia.”